Pathology of the blood group system and transfusion medicine focuses on blood typing, antigen–antibody reactions, compatibility testing, and transfusion reactions. Essential for understanding safe blood administration, laboratory diagnostics, and evidence‑based nursing and medical practice.
Introduction
Blood group systems and transfusion medicine constitute a critical domain within haematology, influencing patient care in surgery, trauma, obstetrics, and transplantation. Understanding the molecular and genetic underpinnings of blood groups, their clinical significance, and the principles guiding safe transfusion practices is essential for medical professionals.
Blood Group Systems
ABO Blood Group System
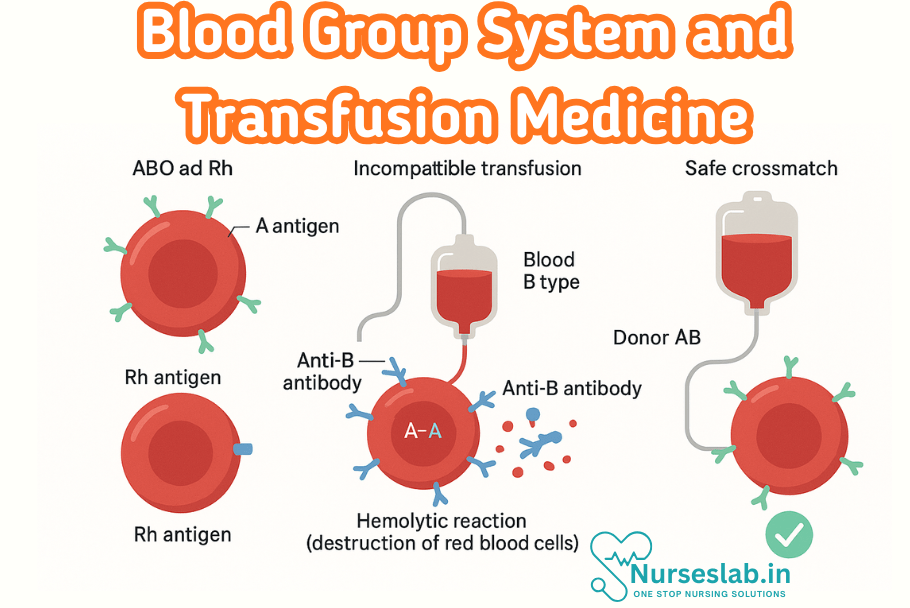
The ABO blood group system, discovered by Karl Landsteiner in 1901, remains the most clinically significant. The system is determined by the presence or absence of A and B antigens on the surface of red blood cells (RBCs). These antigens are carbohydrate moieties attached to glycoproteins and glycolipids on the erythrocyte membrane.
Genetically, the ABO locus is located on chromosome 9 (9q34). It encodes glycosyltransferase enzymes that modify the H antigen, producing either the A or B antigen. The O allele is characterised by an inactive enzyme, resulting in the unmodified H antigen. Individuals inherit one allele from each parent, leading to four possible phenotypes: A, B, AB, and O. The corresponding antibodies (isoagglutinins) in plasma are naturally occurring and are crucial in transfusion compatibility.
Rh Blood Group System
The Rh system, particularly the D antigen, is the second most significant in clinical practice. The presence (Rh-positive) or absence (Rh-negative) of the D antigen on RBCs is determined by the RHD gene on chromosome 1 (1p36.11). The RHCE gene encodes other antigens (C, c, E, e), contributing to the complexity of the system.
Rh antigens are proteinaceous, and their immunogenicity is high; exposure of an Rh-negative individual to Rh-positive blood can result in alloimmunisation, with severe consequences in subsequent transfusions or pregnancies.
Minor Blood Group Systems
Several other blood group systems have clinical relevance, especially in patients receiving multiple transfusions or those with rare blood types:
- Kell System: The Kell antigen (K) is highly immunogenic and can cause haemolytic transfusion reactions and haemolytic disease of the foetus and newborn (HDFN).
- Duffy System (Fy): Duffy antigens serve as receptors for Plasmodium vivax and are significant in malaria resistance.
- Kidd System (Jk): Kidd antibodies are notorious for causing delayed haemolytic transfusion reactions.
- MNS, Lutheran, Lewis, P: These systems, while less commonly implicated, can be important in specific clinical situations.
Each blood group system is defined by genes encoding specific proteins or carbohydrates on the RBC membrane, contributing to the diversity and complexity of transfusion medicine.
Clinical Significance of Blood Group Systems
The clinical importance of blood group antigens lies in their role in transfusion compatibility, organ transplantation, and disease associations. Incorrect blood group matching can result in acute or delayed transfusion reactions, which may be life-threatening.
- Transfusion Medicine: Accurate determination of ABO and Rh status is mandatory before blood transfusion. Minor antigens become relevant in patients with multiple transfusions, such as those with thalassaemia or sickle cell anaemia.
- Transplantation: ABO compatibility is critical in solid organ transplantation, particularly renal and cardiac transplants. Incompatible transplantation can lead to hyperacute rejection.
- Disease Associations: Certain blood groups are associated with disease susceptibility. For example, individuals with blood group O have a lower risk of coronary artery disease, while those with Duffy negativity (Fy(a-b-)) are resistant to P. vivax malaria.
Pathology of Blood Groups
Haemolytic Disease of the Foetus and Newborn (HDFN)
HDFN arises when maternal antibodies cross the placenta and target foetal RBC antigens inherited from the father. The most common causes are anti-D (Rh incompatibility) and, less frequently, anti-Kell and anti-c antibodies. HDFN can result in foetal anaemia, hydrops foetalis, and even intrauterine death.
Prevention of Rh alloimmunisation, primarily through anti-D immunoglobulin administration to Rh-negative mothers, has significantly decreased the incidence and severity of HDFN.
Autoimmune Haemolytic Anaemia (AIHA)
AIHA occurs when autoantibodies target self-RBC antigens, leading to their destruction. Warm AIHA is mediated by IgG and typically targets Rh antigens, while cold agglutinin disease involves IgM antibodies against I or i antigens. The underlying causes may be idiopathic, secondary to autoimmune disorders, infections, or malignancy.
Alloimmune Haemolytic Transfusion Reactions
Alloimmunisation may occur in transfusion recipients exposed to foreign RBC antigens. The immune response can lead to acute or delayed haemolytic transfusion reactions, depending on the antibody class and antigen involved.
- Acute reactions: Often due to ABO incompatibility, leading to intravascular haemolysis, renal failure, and disseminated intravascular coagulation.
- Delayed reactions: Frequently caused by minor antigens (Kidd, Duffy, Kell), manifesting several days post-transfusion with mild jaundice and anaemia.
Rare Blood Group Disorders
Certain rare blood group phenotypes, such as the Bombay (Oh) phenotype, lack the H antigen and are incompatible with all standard ABO blood types. Such individuals require blood from similarly rare donors, posing significant challenges in transfusion support.
Other rare conditions include McLeod syndrome (associated with Kell system), and Gerbich negativity (linked to the Gerbich system). These rare phenotypes may be associated with unique clinical syndromes and transfusion requirements.
Principles of Transfusion Medicine
Blood Component Therapy
Modern transfusion medicine emphasises the use of specific blood components rather than whole blood, optimising therapy and minimising risks. Key components include:
- Red Blood Cell Concentrates: Used to treat anaemia and acute blood loss.
- Platelet Concentrates: Indicated for thrombocytopaenia or platelet dysfunction.
- Fresh Frozen Plasma (FFP): Contains clotting factors, used in coagulopathies.
- Cryoprecipitate: Rich in fibrinogen and factor VIII, used in haemophilia and hypofibrinogenaemia.
Each component is prepared, stored, and administered according to stringent protocols to preserve efficacy and safety.
Donor Selection and Blood Collection
Donor selection criteria prioritise safety for both donor and recipient. Screening involves medical history, physical examination, and laboratory testing for infectious diseases (HIV, HBV, HCV, syphilis, malaria). Blood is collected aseptically, with appropriate labelling and traceability to ensure accountability.
Storage and Handling
Blood components require specific storage conditions:
- RBCs: 2–6°C, up to 42 days with additive solutions.
- Platelets: 20–24°C with gentle agitation, for up to 5–7 days.
- Plasma and cryoprecipitate: ≤−18°C, up to one year.
Proper storage minimises the risk of bacterial proliferation, degradation of components, and ensures maximum therapeutic benefit.
Compatibility Testing in Transfusion Medicine
Principles of Compatibility Testing
Compatibility testing is crucial to prevent transfusion reactions. It involves:
- ABO and Rh Typing: Determining the patient’s and donor’s blood groups.
- Antibody Screening: Detects unexpected antibodies in the recipient’s serum against donor RBC antigens.
- Crossmatching: Combines recipient serum with donor RBCs to assess compatibility. Immediate spin and indirect antiglobulin (Coombs) tests are employed.
These tests are performed prior to each transfusion episode, with protocols varying based on the urgency and clinical context.
Pre-Transfusion Protocols
Pre-transfusion protocols include:
- Patient identification and labelling of samples.
- Verification of blood component type, expiry, and compatibility report.
- Documentation of indication and informed consent.
- Baseline vital signs before transfusion.
Strict adherence to these protocols reduces the risk of human error and adverse events.
Transfusion Reactions: Types, Mechanisms, and Management
Acute Transfusion Reactions
- Acute Haemolytic Reaction: Most often due to ABO incompatibility. Presents with fever, chills, back pain, hypotension, and haemoglobinuria. Immediate cessation of transfusion and supportive management are essential.
- Febrile Non-Haemolytic Reaction: Caused by cytokines or recipient antibodies to donor leukocytes. Manifests as fever and chills without haemolysis. Treated symptomatically.
- Allergic Reactions: Range from mild urticaria to severe anaphylaxis, often due to plasma proteins. Management includes antihistamines and, in severe cases, adrenaline.
- Transfusion-Related Acute Lung Injury (TRALI): Characterised by acute respiratory distress within 6 hours of transfusion, caused by donor anti-leukocyte antibodies. Requires supportive respiratory care.
- Transfusion-Associated Circulatory Overload (TACO): Presents with hypertension, pulmonary oedema, and heart failure in susceptible patients. Managed by diuretics and slowing or stopping the transfusion.
Delayed Transfusion Reactions
- Delayed Haemolytic Reaction: Occurs days to weeks post-transfusion, often due to Kidd, Duffy, or Kell antibodies. Presents as unexplained anaemia and jaundice.
- Post-Transfusion Purpura: Rare; caused by alloantibodies against platelet antigens, leading to severe thrombocytopaenia.
- Transfusion-Transmitted Infections: Despite rigorous screening, there remains a small risk of transmission of viruses, bacteria, or parasites.
Clinical Management of Transfusion Reactions
Management involves immediate cessation of the transfusion, assessment of the patient, supportive measures, and notification of transfusion services for investigation. Retrospective laboratory testing is performed to confirm diagnosis and implement corrective actions.
Safety and Protocols in Transfusion Medicine
Infection Prevention
Stringent donor screening and laboratory testing are the mainstays of infection prevention. Pathogen inactivation technologies are increasingly employed to further reduce risk.
Adverse Event Reporting
All transfusion-related adverse events must be documented and reported according to institutional and regulatory guidelines. Haemovigilance programmes monitor trends and facilitate continuous quality improvement.
Regulatory Standards
Transfusion services in India operate under the Drugs and Cosmetics Act, supervised by agencies such as the National Blood Transfusion Council (NBTC) and the Food and Drug Administration (FDA). Internationally, standards are set by organisations like the World Health Organization (WHO) and the American Association of Blood Banks (AABB).
Recent Advances in Transfusion Medicine
Molecular Typing
Molecular techniques, such as polymerase chain reaction (PCR) and next-generation sequencing, now enable precise blood group genotyping beyond serological methods. These advances assist in identifying rare phenotypes and managing complex alloimmunised patients.
Pathogen Reduction Technologies
Innovative methods, such as photochemical treatment and solvent-detergent processes, reduce the risk of transfusion-transmitted infections, particularly in plasma and platelets.
Emerging Technologies
Automated blood grouping platforms, artificial intelligence for donor-recipient matching, and the development of universal donor RBCs through enzymatic modification of antigens represent the future frontiers of transfusion medicine.
REFERENCES
- Ramadas Nayak, Textbook of Pathology and Genetics for Nurses, 2nd Edition,2024, Jaypee Publishers, ISBN: 978-93-5270-031-8.
- Suresh Sharma, Textbook of Pharmacology, Pathology & Genetics for Nurses II, 2nd Edition, 31 August 2022, Jaypee Publishers, ISBN: 978-9354655692.
- Kumar, V., Abbas, A.K., & Aster, J.C. (2020). Robbins and Cotran Pathologic Basis of Disease. 10th Edition. Elsevier.
- McCance, K.L., & Huether, S.E. (2018). Pathophysiology: The Biologic Basis for Disease in Adults and Children. 8th Edition. Elsevier.
- McCance KL, Huether SE. Pathophysiology: the biologic basis for disease in adults and children, 8th edn. St Louis (MI): Mosby; 2018, https://www.britishjournalofnursing.com/content/clinical-new-series/pathophysiology-applied-to-nursing-the-basis-for-disease-and-illness
Stories are the threads that bind us; through them, we understand each other, grow, and heal.
JOHN NOORD
Connect with “Nurses Lab Editorial Team”
I hope you found this information helpful. Do you have any questions or comments? Kindly write in comments section. Subscribe the Blog with your email so you can stay updated on upcoming events and the latest articles.





