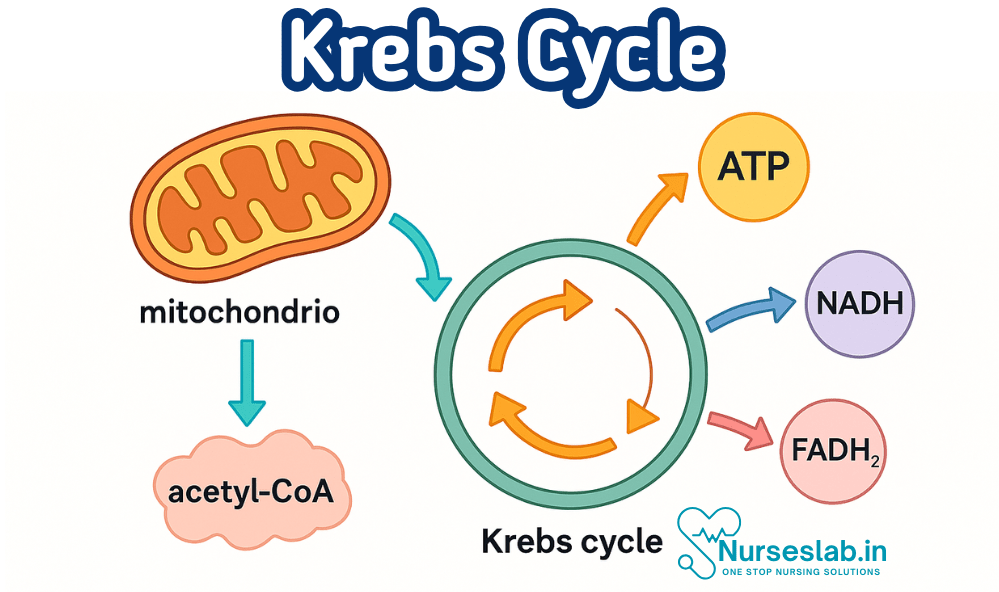
The Krebs cycle, or citric acid cycle, occurs in mitochondria and oxidizes acetyl-CoA to generate ATP, NADH, and FADH₂. This process supports energy production, metabolism, and cellular respiration—essential in physiology, nursing, and medical education.
Introduction
The Krebs Cycle, also known as the Citric Acid Cycle or the Tricarboxylic Acid (TCA) Cycle, is a series of chemical reactions that occur in the mitochondria of cells. Its primary function is to generate energy through the oxidation of acetyl-CoA derived from carbohydrates, fats, and proteins. The energy produced is stored in molecules such as ATP (adenosine triphosphate), which cells use to perform essential functions.
Historical Background:
The Krebs Cycle was first described by Sir Hans Adolf Krebs in 1937, earning him the Nobel Prize in Physiology or Medicine in 1953. His discovery revealed how living organisms extract energy from food, a breakthrough that transformed our understanding of metabolism. The cycle’s significance extends beyond biochemistry; it provides a framework for understanding metabolic diseases, the effects of drugs, and the body’s response to injury and illness.
Overview of Cellular Respiration
Cellular respiration is the process by which cells harvest energy from nutrients. It consists of three main stages:
- Glycolysis: The breakdown of glucose into pyruvate in the cytoplasm, generating small amounts of ATP and NADH.
- Krebs Cycle: The oxidation of acetyl-CoA in the mitochondrial matrix, producing NADH, FADH2, and CO2.
- Electron Transport Chain (ETC): The transfer of electrons from NADH and FADH2 to oxygen, producing the majority of cellular ATP.
The Krebs Cycle sits at the heart of metabolism, linking the breakdown of carbohydrates, fats, and proteins to energy production. It also provides intermediates for other biosynthetic processes, making it a hub for metabolic activity.
Detailed Steps of the Krebs Cycle
Chemical Reactions, Enzymes Involved, and Key Intermediates
The Krebs Cycle consists of eight main steps, each catalysed by a specific enzyme. The cycle begins with the condensation of acetyl-CoA and oxaloacetate to form citrate, and ends with the regeneration of oxaloacetate. Here is a step-by-step breakdown:
- Condensation of Acetyl-CoA and Oxaloacetate (Citrate Synthase): Acetyl-CoA (2 carbons) combines with oxaloacetate (4 carbons) to form citrate (6 carbons). This reaction is catalysed by citrate synthase and is highly exergonic, helping to drive the cycle forward.
- Isomerisation of Citrate to Isocitrate (Aconitase): Citrate is rearranged to isocitrate through the intermediate cis-aconitate, catalysed by the enzyme aconitase. This prepares the molecule for subsequent oxidation.
- Oxidative Decarboxylation of Isocitrate (Isocitrate Dehydrogenase): Isocitrate is oxidised and decarboxylated to form α-ketoglutarate (5 carbons), releasing one molecule of CO2 and reducing NAD+ to NADH. Isocitrate dehydrogenase is a key regulatory enzyme in the cycle.
- Oxidative Decarboxylation of α-Ketoglutarate (α-Ketoglutarate Dehydrogenase): α-Ketoglutarate is further oxidised and decarboxylated to form succinyl-CoA (4 carbons), with the release of another CO2 and the reduction of another NAD+ to NADH. This step is catalysed by the α-ketoglutarate dehydrogenase complex.
- Conversion of Succinyl-CoA to Succinate (Succinyl-CoA Synthetase): Succinyl-CoA is converted to succinate, catalysed by succinyl-CoA synthetase. This reaction generates GTP (guanosine triphosphate), which can be readily converted to ATP.
- Oxidation of Succinate to Fumarate (Succinate Dehydrogenase): Succinate is oxidised to fumarate, with the reduction of FAD (flavin adenine dinucleotide) to FADH2. Succinate dehydrogenase is notable for being embedded in the inner mitochondrial membrane and also functions as part of the electron transport chain.
- Hydration of Fumarate to Malate (Fumarase): Fumarate is hydrated to malate by the enzyme fumarase.
- Oxidation of Malate to Oxaloacetate (Malate Dehydrogenase): Malate is oxidised to regenerate oxaloacetate, with the reduction of NAD+ to NADH. This completes the cycle, allowing it to begin again with a new acetyl-CoA molecule.
Summary Table: Key Steps, Enzymes, and Products
| Step | Enzyme | Substrate(s) | Product(s) | Energy Carrier Produced |
| 1 | Citrate Synthase | Acetyl-CoA + Oxaloacetate | Citrate | – |
| 2 | Aconitase | Citrate | Isocitrate | – |
| 3 | Isocitrate Dehydrogenase | Isocitrate | α-Ketoglutarate + CO<sub>2</sub> | NADH |
| 4 | α-Ketoglutarate Dehydrogenase | α-Ketoglutarate | Succinyl-CoA + CO<sub>2</sub> | NADH |
| 5 | Succinyl-CoA Synthetase | Succinyl-CoA | Succinate | GTP (→ ATP) |
| 6 | Succinate Dehydrogenase | Succinate | Fumarate | FADH<sub>2</sub> |
| 7 | Fumarase | Fumarate | Malate | – |
| 8 | Malate Dehydrogenase | Malate | Oxaloacetate | NADH |
Energy Yield and Products: ATP, NADH, FADH2, CO2
Each turn of the Krebs Cycle produces the following energy carriers and by-products, which are essential for cellular function:
- 3 NADH: Each NADH carries high-energy electrons to the electron transport chain, leading to the production of ATP.
- 1 FADH2: Also donates electrons to the electron transport chain, producing ATP.
- 1 GTP (equivalent to ATP): Directly usable by the cell for energy-dependent processes.
- 2 CO2: Waste products expelled during respiration.
Since each glucose molecule yields two acetyl-CoA molecules, the cycle turns twice for each glucose, effectively doubling these yields per glucose molecule.
Regulation of the Krebs Cycle
The Krebs Cycle is tightly regulated to meet the cell’s energy demands and maintain metabolic balance. The main regulatory mechanisms involve:
- Enzyme Regulation: The three key regulatory enzymes are citrate synthase, isocitrate dehydrogenase, and α-ketoglutarate dehydrogenase. These enzymes are sensitive to the levels of ATP, NADH, and other metabolites.
- Energy Status: High levels of ATP and NADH indicate sufficient energy, inhibiting the cycle. Conversely, low ATP/ADP ratios stimulate the cycle to produce more energy.
- Substrate Availability: The availability of acetyl-CoA, oxaloacetate, and NAD+ influences cycle activity.
- Calcium Ions: Calcium, released during muscle contraction, can activate certain enzymes in the cycle, linking energy production to muscle activity.
Understanding these control points helps nurses appreciate how metabolic pathways adjust during exercise, stress, fasting, or illness.
Clinical Relevance
Inherited Metabolic Disorders
Several rare genetic disorders affect enzymes of the Krebs Cycle, leading to severe metabolic consequences. Examples include:
- Fumarase Deficiency: Causes developmental delay, encephalopathy, and seizures due to impaired energy production in the brain.
- Succinate Dehydrogenase Deficiency: Associated with mitochondrial diseases, muscle weakness, and neurological symptoms.
- α-Ketoglutarate Dehydrogenase Deficiency: Leads to lactic acidosis and neurological dysfunction.
Acquired Conditions
Krebs Cycle dysfunction can also occur in acquired conditions such as:
- Ischaemia (Reduced Blood Flow): Oxygen deprivation impairs the electron transport chain, causing a backup of NADH and inhibiting the Krebs Cycle. This is seen in stroke, myocardial infarction, and shock.
- Vitamin Deficiencies: Several vitamins (e.g., niacin, riboflavin, thiamine) are cofactors for Krebs Cycle enzymes. Deficiencies can impair energy production and contribute to clinical symptoms such as fatigue, muscle weakness, and neurological changes.
- Poisoning: Certain toxins, such as arsenic, can inhibit Krebs Cycle enzymes, leading to metabolic failure.
Diagnostic Implications
Disorders of the Krebs Cycle may present with non-specific symptoms such as fatigue, muscle pain, and neurological disturbances. Laboratory findings may include lactic acidosis, elevated levels of certain organic acids in urine, and abnormal mitochondrial function tests. Understanding the biochemical basis of these conditions aids in diagnosis and management, allowing nurses to contribute meaningfully to patient assessment and care planning.
Implications for Patient Care
A sound knowledge of the Krebs Cycle has practical implications for nursing care. Key considerations include:
- Recognising Signs of Metabolic Disturbance: Nurses should be alert to symptoms such as unexplained fatigue, muscle weakness, or altered mental status, which may indicate underlying metabolic dysfunction.
- Supporting Nutritional Needs: Patients with increased metabolic demands (e.g., post-surgery, trauma, sepsis) require adequate nutrition to support the Krebs Cycle and overall energy production. Ensuring sufficient intake of vitamins and minerals is crucial for enzyme function.
- Monitoring for Drug and Toxin Effects: Some medications and toxins can interfere with the Krebs Cycle. Nurses must monitor patients for adverse effects and report changes promptly.
- Patient Education: Nurses play a vital role in educating patients about the importance of balanced nutrition, the effects of certain drugs or toxins, and recognising early signs of metabolic imbalance.
- Managing Acutely Ill Patients: In critical care settings, understanding the interplay between oxygen delivery, energy production, and metabolic waste helps nurses anticipate complications and tailor interventions.
Connections to Other Metabolic Pathways
Link to Glycolysis
The Krebs Cycle is intimately connected to glycolysis, the process by which glucose is broken down in the cytoplasm to produce pyruvate. Pyruvate enters the mitochondria and is converted to acetyl-CoA, the substrate for the Krebs Cycle. In situations where oxygen is limited (anaerobic conditions), pyruvate is instead converted to lactate, bypassing the Krebs Cycle and leading to less efficient energy production.
Link to the Electron Transport Chain (ETC)
The NADH and FADH2 generated by the Krebs Cycle carry high-energy electrons to the ETC, where most ATP is produced. The ETC requires oxygen as the final electron acceptor; without it, the cycle slows down and energy production decreases. This connection explains why oxygen delivery is so critical in patient care, especially in respiratory or circulatory compromise.
Link to Fatty Acid Metabolism
Fatty acids are broken down via β-oxidation to produce acetyl-CoA, which then enters the Krebs Cycle. During fasting or prolonged exercise, the body shifts from using glucose to fatty acids for energy, increasing reliance on the Krebs Cycle for ATP production. Disorders affecting fatty acid metabolism can therefore impact the Krebs Cycle and overall energy status.
Amino Acid Metabolism
Some amino acids can be converted into Krebs Cycle intermediates, linking protein metabolism to energy production. In conditions of increased protein breakdown (e.g., severe illness, trauma), amino acids contribute to maintaining energy balance via the cycle.
Summary and Key Takeaways
- The Krebs Cycle is central to energy production in all cells, converting nutrients into ATP, the body’s energy currency.
- It consists of eight steps, each catalysed by a specific enzyme, producing NADH, FADH2, GTP (ATP), and CO2.
- Regulation of the cycle ensures energy production matches demand. Disruptions can lead to metabolic disorders with clinical implications.
- Nurses must be aware of conditions affecting the Krebs Cycle, such as inherited metabolic diseases, vitamin deficiencies, and effects of drugs or toxins.
- Understanding the Krebs Cycle enhances patient assessment, supports nutritional and pharmacological interventions, and underpins effective education.
- The cycle connects with glycolysis, the electron transport chain, and fatty acid and amino acid metabolism, forming the backbone of cellular energetics.
REFERENCES
- Harbans Lal, Textbook of Applied Biochemistry and Nutrition& Dietetics 2nd Edition ,November 2024, CBS Publishers and Distributors, ISBN: 978-9394525757
- Suresh K Sharma, Textbook of Biochemistry and Biophysics for Nurses, 2nd Edition, September 2022, Jaypee Publishers, ISBN: 978-9354655760
- Peter J Kennelly, Harpers Illustrated Biochemistry Standard Edition, September 2022, McGraw Hill Lange Publishers, ISBN: 978-1264795673
- Denise R Ferrier, Ritu Singh, Lippincott Illustrated Reviews Biochemistry, Second Edition, June 2024, ISBN- 978-8197055973
- Yadav, Tapeshwar & Bhadeshwar, Sushma. (2022). Essential Textbook of Biochemistry for Nursing.
- Applied Sciences, Importance of Biochemistry for Nursing Practice, November 2, 2023, https://bns.institute/applied-sciences/importance-biochemistry-nursing-practice/
Stories are the threads that bind us; through them, we understand each other, grow, and heal.
JOHN NOORD
Connect with “Nurses Lab Editorial Team”
I hope you found this information helpful. Do you have any questions or comments? Kindly write in comments section. Subscribe the Blog with your email so you can stay updated on upcoming events and the latest articles.





